How Do Electronegativity And Polarity Relate
Chemical bonding Electronegativity table periodic bond chemistry chart polarity general pauling values chemical elements energy ionization principles element trends applications patterns scale Bond electronegativity polarity polar covalent ionic bonding libretexts chemistry map nonpolar maps general
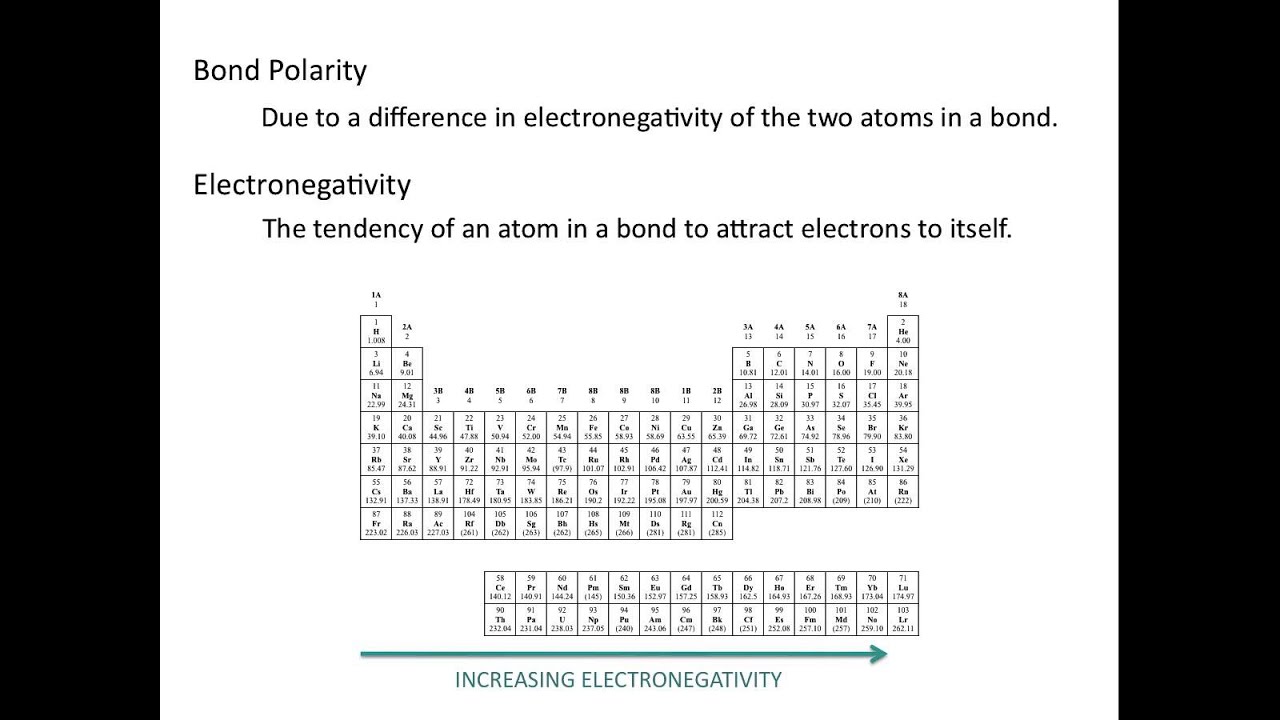
What trend in electronegativity do you see as you go across a period
Electronegativity polarity tumblr Electronegativity polar covalent bonds compounds explain differences Electronegativity and polarity
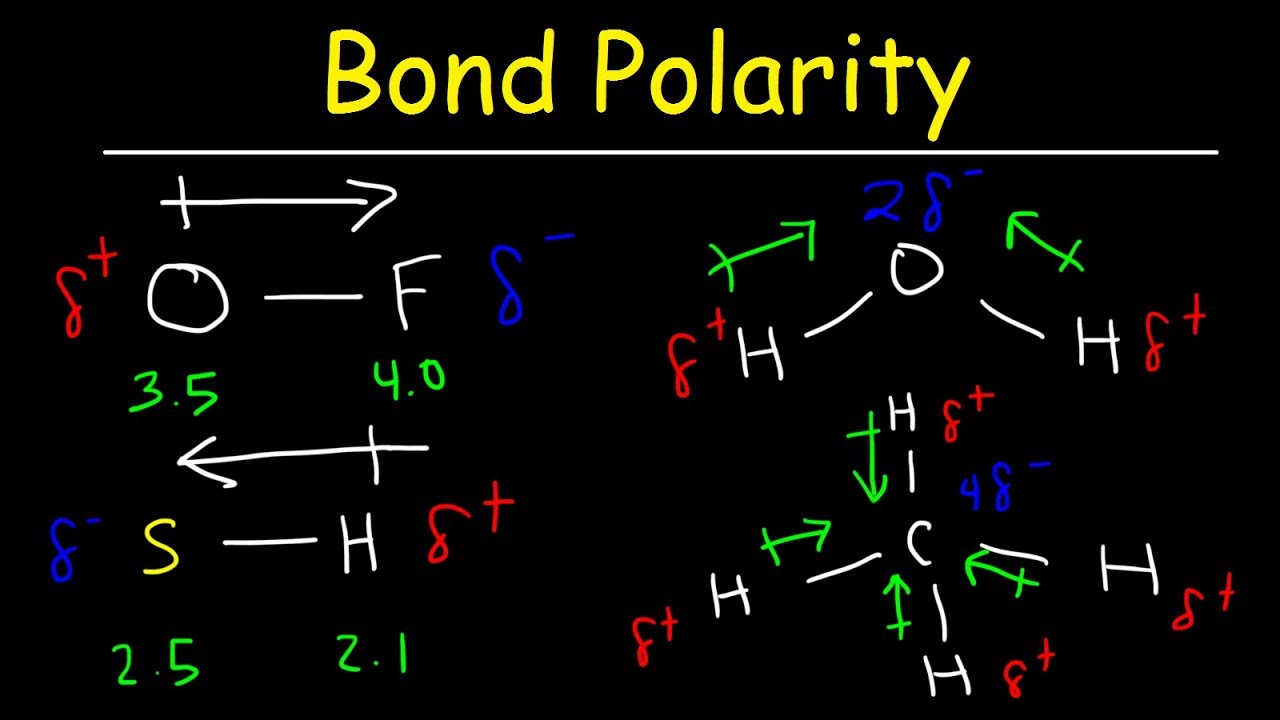
8.4: bond polarity and electronegativity
8.4: bond polarity and electronegativityWhich atom in each pair that has the greater electronegativity. a. ca Polarity bond dipole electronegativity moment chemistry practice problemsWhat trend in electronegativity do you see as you go across a period.
Trends electronegativity periodic ionization energy table chart has electronegativities electrons describes another here videoStoichiometric basics: chemistry for kids! Here is another video that describes ionization energy trends in theElectronegativity and bond polarity.

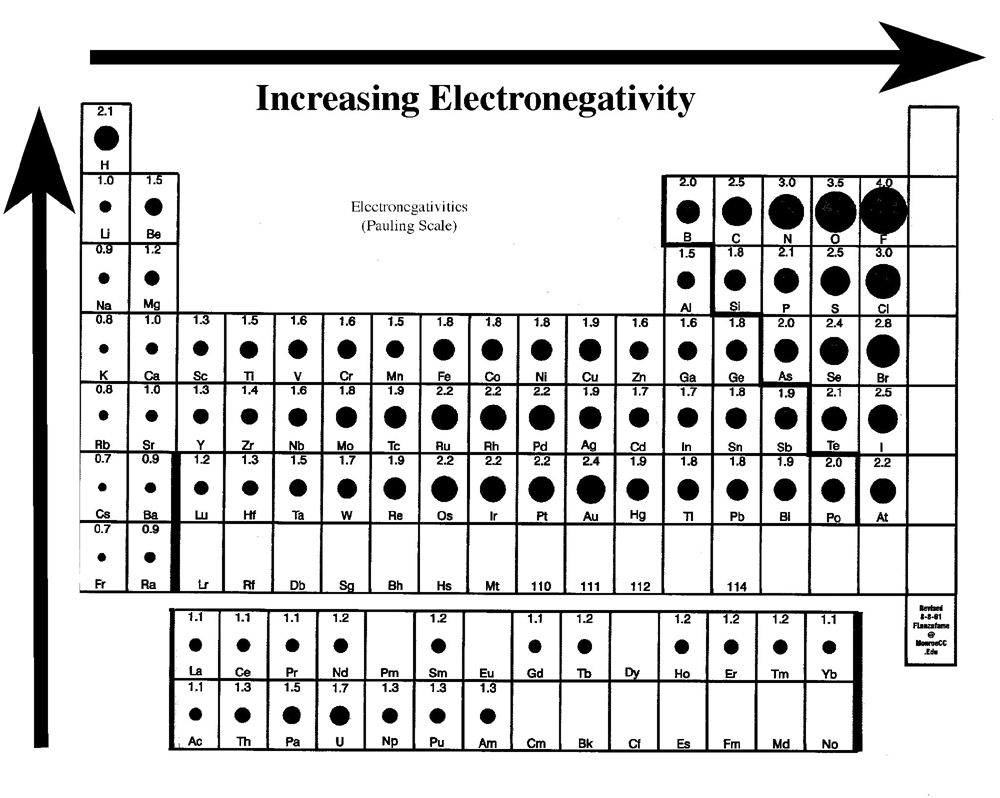
Electronegativity oxidation table number chemistry introduction lowest highest elements bottom left right top
Electronegativity differences explain polar bonds in covalent compoundsHow can i determine bond polarity? + example Electronegativity chart polarity periodic elements table type bond difference charts element determine atoms chemistry two electronegative most atom trends commonBond chemistry electronegativity ionic binding verschil covalente covalent teaching polar bonding table polaire een gradation periodic kids choose board.
Electronegativity periodic trends bonding chemical trend chart element polarity bond electrons tendency atom electronegative table increasing electron attraction chemistry attractNonpolar bonds electronegativity hydrogen biology oxygen electronegative fluorine nitrogen chemistry most overview sylvia freeman Electronegativity periodic atom atomic pair covalent electronegative ionic radius chloride atoms greater electrons ions values increases columnsElectronegativity and oxidation number.

Electronegativity periodic trend electron period affinity table go across chemistry trends behavior electronegativities presentation row sliderbase do increase when decrease
Polar vs. nonpolar bonds — overview & examplesBond polarity, electronegativity and dipole moment Polarity electronegativity bond chemistry.
.